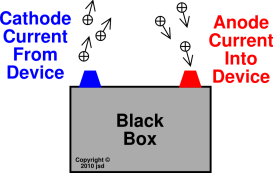
It is really important that you get these the right way round! The negative electrode is called the cathode. The positive electrode is called the anode. Most of the time in the lab, you use carbon (graphite) electrodes, but occasionally you might come across platinum being used. In most cases you will come across, these aren't changed by the electrolysis, and we describe them as "inert". The electrodes are the pieces of metal or carbon I have shown as black rods in the diagram above. Using this simple circuit for electrolysis After the small virtually instantaneous movement of electrons at the beginning, there is no further current flow. If you put a battery or ammeter into the circuit, nothing would happen to either. Eventually, the incoming electrons just get repelled by the ones already there and there is no further increase in negativity.Įlectrons are beng pumped away from the right-hand side, and that shortage of electrons leaves it positively charged. As you pump more electrons on to the left-hand piece of metal in the diagram, it becomes negatively charged. How many electrons get pumped depends on the strength of the power source. The power source will pump electrons from one to piece to the other. Suppose you connected two bits of metal or carbon to a direct current (dc) power source of some kind. The simplest way of thinking about a battery or other similar power source is as an electron pump. If you want a description of this structure you could look at this page. In a carbon (graphite) electrode, there are also delocalised electrons which can move through the structure. If you aren't confident about metallic bonding then have a look at the metallic bonding page before you go on. In a metal, the electrons flowing are the delocalised electrons (also known as a "sea of electrons") which hold the metal together. You will find detailed examples of electrolysis in other pages in this section.Įlectrolysis is breaking up an ionic compound (either molten or in solution) into simpler substances by using an electric current.Ī flow of electricity through a metal or carbon is simply a flow of electrons. This page introduces the key words and ideas in electrolysis.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
